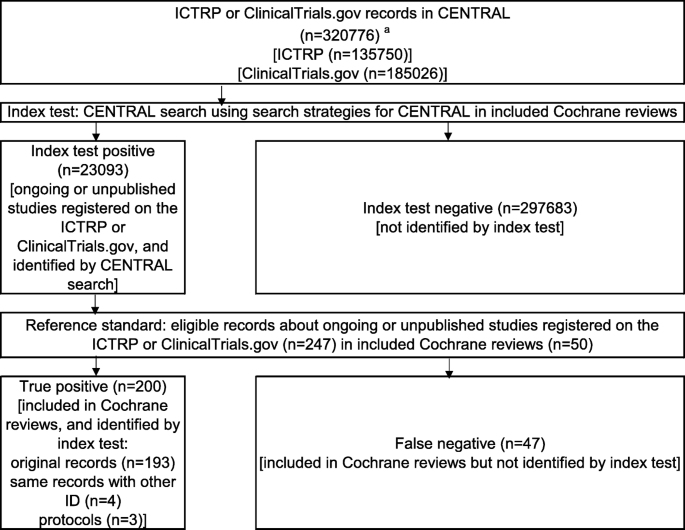
Number of clinical trial registrations in each CTR, percentage of those... | Download Scientific Diagram
PLOS ONE: Prevalence of clinical trial status discrepancies: A cross-sectional study of 10,492 trials registered on both ClinicalTrials.gov and the European Union Clinical Trials Register

Ongoing Clinical Trials for the Management of the COVID-19 Pandemic: Trends in Pharmacological Sciences

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet

The Exclusion of Patients with CKD in Prospectively Registered Interventional Trials for COVID-19—a Rapid Review of International Registry Data | American Society of Nephrology

Prospective registration and reporting of trial number in randomised clinical trials: global cross sectional study of the adoption of ICMJE and Declaration of Helsinki recommendations | The BMJ